Imricor pushes NorthStar deeper into the US market with pediatric FDA filing
Imricor Medical Systems has taken another step in its US rollout, lodging its NorthStar Mapping System with the FDA for a pediatric label expansion via the Special 510(k) pathway. For investors, the key point is not just the filing itself, but what it says about the company’s commercial playbook: get NorthStar into receptive hospitals early, build an installed base, and use that foothold to support the broader launch of its full electrophysiology platform in the world’s biggest healthcare market.
A logical follow-up to January’s adult clearance
The move follows NorthStar’s FDA clearance for adult patients in January 2026, so this is not a standing-start regulatory event. Rather, it looks like an extension of a strategy already under way. Imricor is trying to convert regulatory progress into actual hospital adoption, and the pediatric segment appears to be one of the first commercial doors it believes it can push open.
That matters because small medtech companies often win by sequencing their markets carefully. Instead of waiting for broad-based adoption across adult hospitals, Imricor is targeting areas where the product’s value proposition may be especially compelling. In this case, that means children’s hospitals, where reducing radiation exposure is likely to resonate strongly with clinicians and administrators alike.
Why pediatrics could be more than a niche
Imricor says there are more than 250 children’s hospitals in the United States and that it has already received inbound interest from that customer group following the adult clearance. That is an encouraging detail because it suggests demand discovery is already happening without the company having to spend heavily to manufacture it. Hospitals are making the running, or at least picking up the phone.
The pediatric angle also appears commercially attractive for another reason. NorthStar is used in interventional cardiovascular magnetic resonance procedures, and Imricor argues it may improve workflows that are currently performed using standard MRI system interfaces alone. If that proves true in practice, the product is not merely a regulatory novelty but a tool that could make an existing clinical process more usable and more efficient. Investors should always pay attention when a device company can point to workflow improvement, because hospitals do not buy technology simply because it is clever. They buy it when it solves a problem.
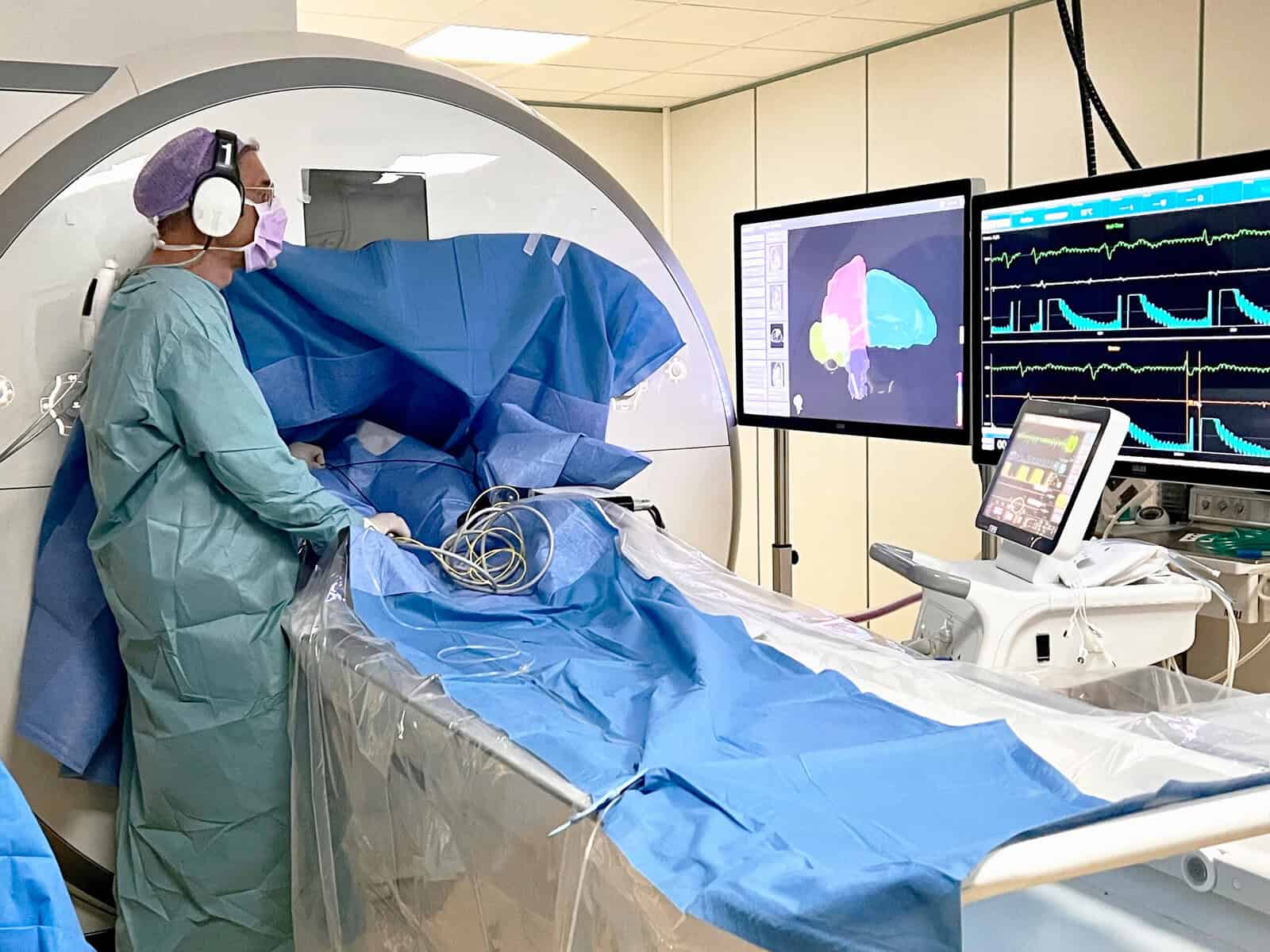
Building revenue before the full platform lands
One of the more interesting aspects of the update is that management frames pediatrics as an early commercial channel ahead of broader US adoption of the full EP platform in adult hospitals. In plain English, Imricor is trying to create revenue opportunities and clinical reference sites now, rather than waiting for the whole strategic puzzle to be completed.
That matters because installed base tends to be one of the most valuable currencies in medtech. Once a hospital adopts a system, trains staff and develops familiarity with its use, the odds improve that further products from the same ecosystem can follow. Imricor is effectively trying to seed the market. A handful of pediatric wins could do more than add sales - they could provide proof points, physician advocates and operational learnings for a wider US expansion.
The company also notes it can already sell NorthStar to children’s hospitals that have proactively contacted it. The label expansion, if cleared, would allow it to market proactively into that segment rather than waiting for inbound interest. That is a meaningful distinction. Passive demand is useful; active selling is better.
Timing, momentum and the next watchpoint
Imricor expects clearance in the current quarter. That gives the market a near-term catalyst to watch. The real test, though, will not be the filing or even the clearance alone. Investors will want to see whether regulatory progress converts into orders, placements, and eventually recurring procedure-related revenue.
Chief executive Steve Wedan said the submission was an “exciting and practical step” to accelerate US commercialisation and argued that pediatric centres could become an important adoption channel. He also used the update to underline a broader strategic message: Imricor does not want to be seen solely as an iCMR ablation company, but as a wider interventional MR business.
That broader framing is important. It suggests NorthStar is not just a single-product story, but part of a larger attempt to establish Imricor in MRI-guided interventional cardiology. For shareholders, that is the attraction and the challenge. The attraction is a potentially differentiated platform in a large market. The challenge is that commercial traction still needs to be demonstrated, one hospital at a time.
For now, the pediatric filing looks like a sensible and potentially value-accretive move. It broadens the addressable market, sharpens the company’s US commercial narrative, and gives investors a clearer sense of how Imricor intends to turn regulatory wins into real-world adoption.